Contact

+86-21-57526862
Tel:+86-21-57526862
Email:
sales@eugenebio.com
Address:No. 1666 Xinyang road, Fengxian district, Shanghai, China
-
Typhoid IgG/IgM
Tel:+86-21-57526862

Packing specification:25T
Intended use:
The EUGENE® Typhoid IgG/IgM Rapid Test is a lateral flow immunochromatographic assay for the simultaneous detection and differentiation of IgG and IgM antibodies to Salmonella typhi (S. typhi) and Paratyphi in human whole blood, serum or plasma. It is intended to be used as an aid in diagnosis of Typhoid fever. This assay provides only a preliminary test results and is recommended for professional use only. All results must be interpreted together with other clinical information available to the physician.
TEST PROCEDURE :
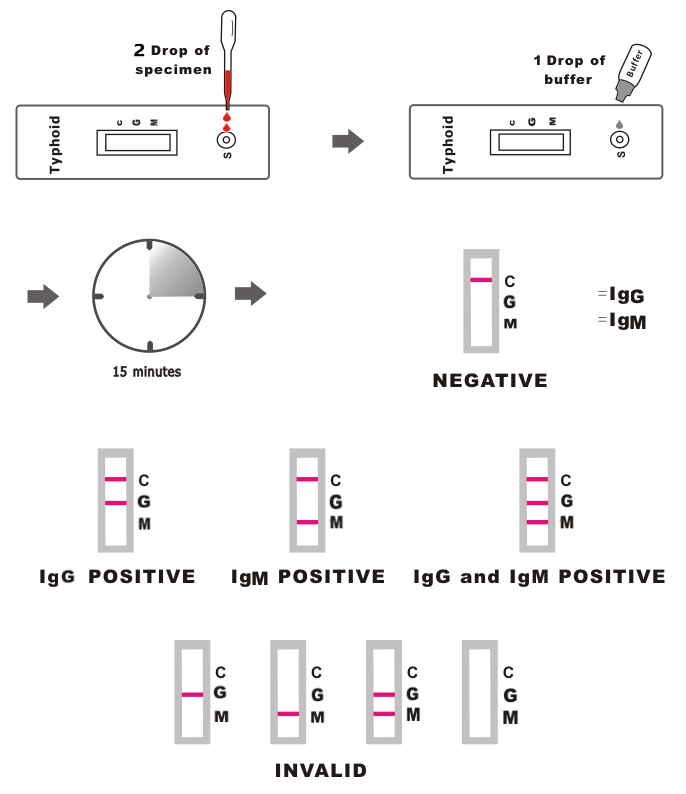
1.Remove the test cassette from the foil pouch; place it on a clean and level surface. Be sure to label the device with specimen’s ID number.
2.Place the test device on a clean and level surface.
For Whole Blood, Serum or Plasma specimens:
Hold the dropper vertically and transfer 2 drops of specimen (or approximately 50 μL) to the specimen well (S) of the test device, then add 1 drop of buffer and start the timer.
For Fingerstick Whole Blood specimens:
To use a capillary tube: Fill the capillary tube and transfer approximately 50 μL (or 2 drops) of fingerstick whole blood specimen to the specimen well (S) of the test device, then add 1 drop of buffer and start the timer.
3.Interpret results at 15-20minutes. Don’t read result after 20 minutes;
Possible setting for the test: Home, workplace, physician’s office or clinical labs
Storage conditions and expiration date:
1. The original package shall be stored at 4℃ ~ 30℃, and shall not be frozen. The validity period is 24 months from the date of verification and qualification.
2. The test paper should be used within 1 hour after the original aluminum foil bag is opened.
 中文版
中文版 English
English